Exploration of larval zebrafish internal states using temperature


Presented by
Guillaume Le Goc
Supervised by
Georges Debrégeas
Raphaël Candelier
October 1st 2021
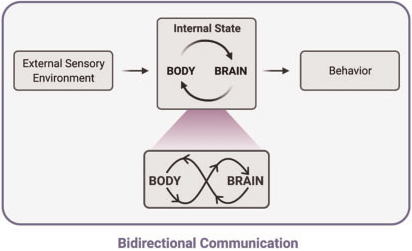
Internal states
"Dynamic, interconnected communication loops distributed across body, brain, and time"
[Kanwal et al., Integr. Comp. Biol. 2021]
Going for a party ?
Yay !
Nope
Mood
All good
Stressed
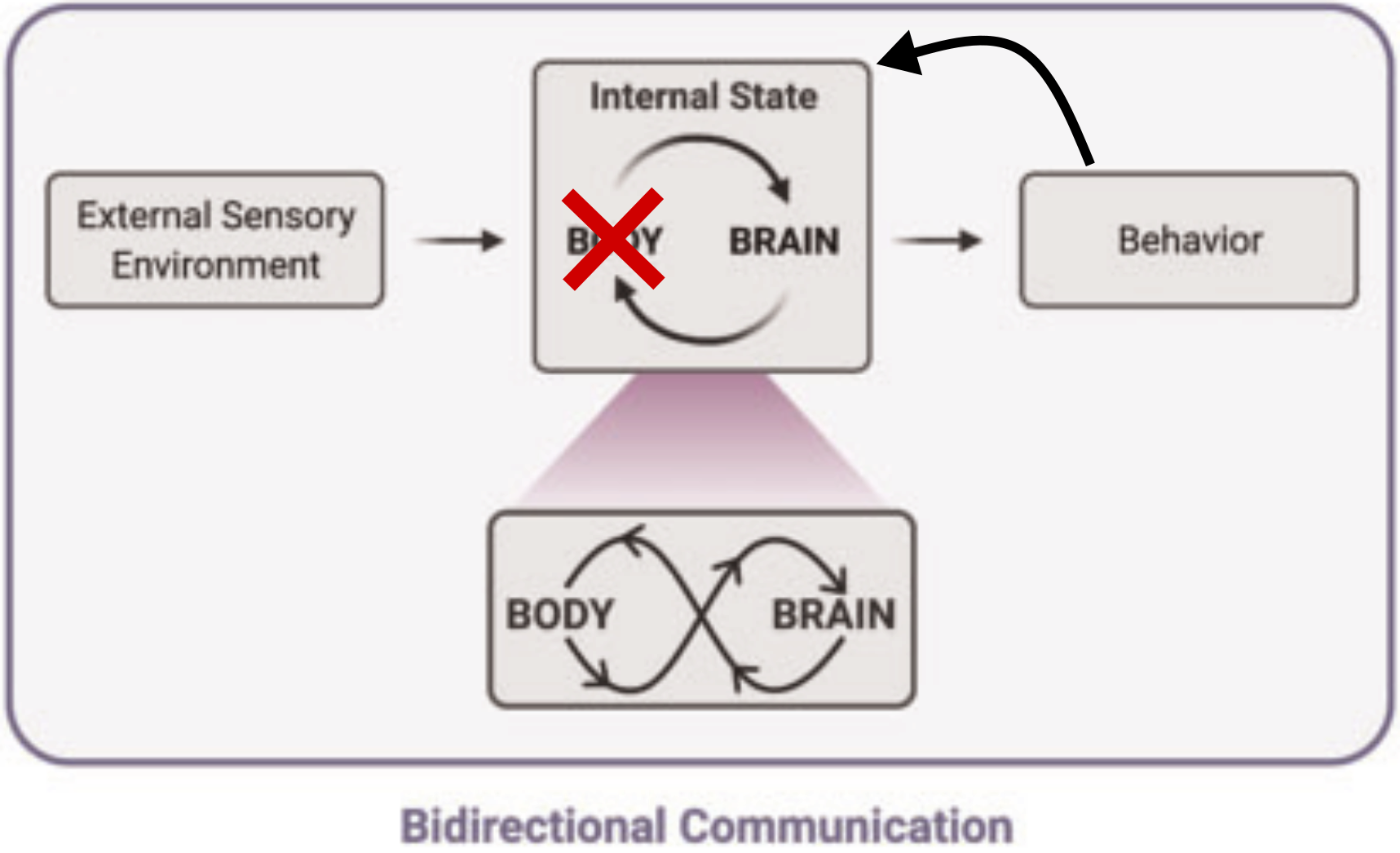
Internal states
"Dynamic, interconnected communication loops distributed across body, brain, and time"
[Kanwal et al., Integr. Comp. Biol. 2021]
Stress
Hunger
Arousal
Fatigue
...
[Kanwal et al., Integr. Comp. Biol. 2021]
Internal states
"Dynamic, interconnected communication loops distributed across body, brain, and time"
[Kanwal et al., Integr. Comp. Biol. 2021]
Stress
Hunger
Arousal
Fatigue
...
[Kanwal et al., Integr. Comp. Biol. 2021]
Model : Zebrafish larva
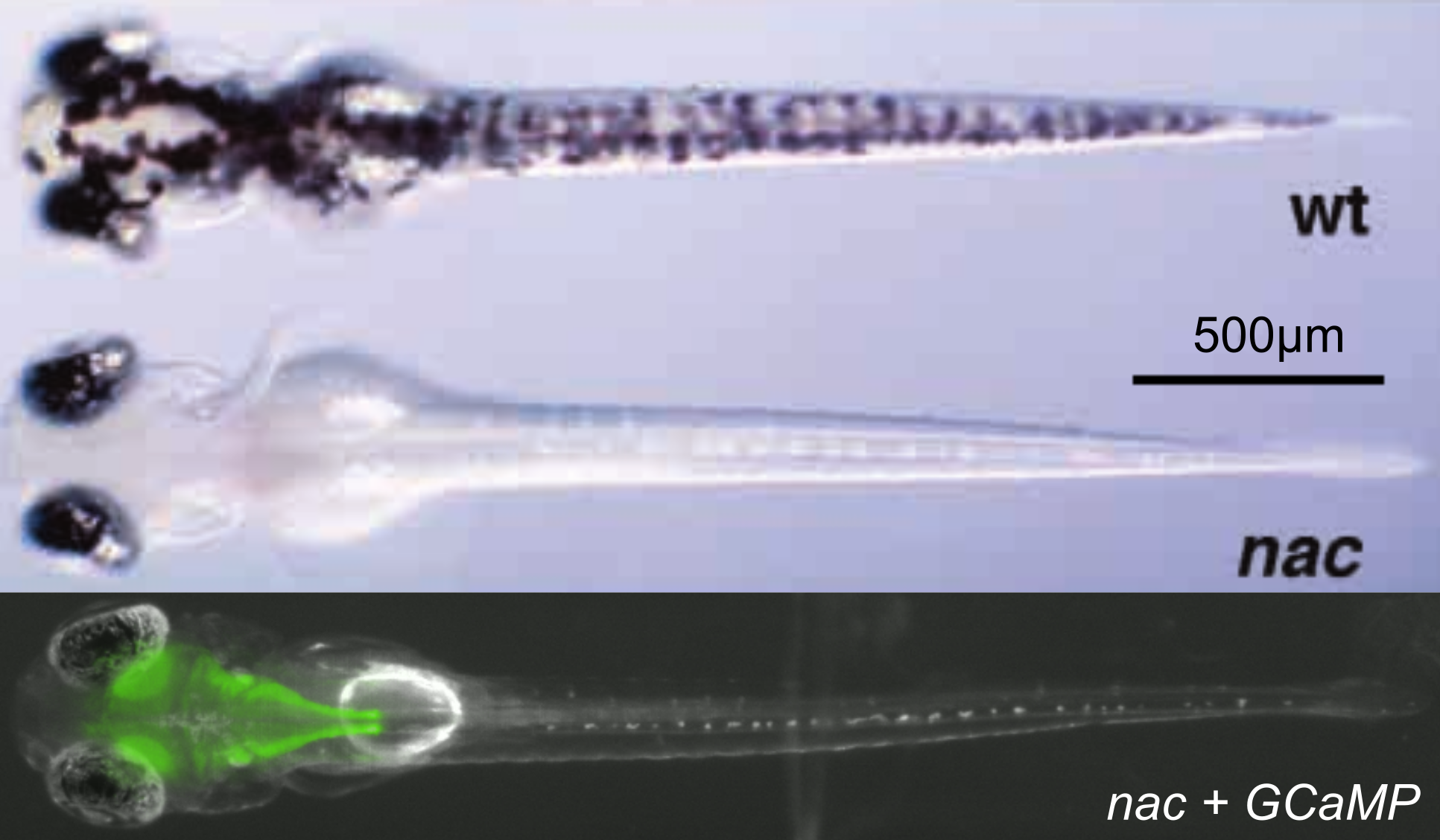
[Lister et. al., Development 1999]
Small & transparent vertebrate,
Mutant & transgenic lines,
Rich behavioral repertoire
Calcium reporter expressed in nearly all neurons : brain-wide activity recordings
[LJP]
100µm
5-7 days post-fertilization larvae
External sensory environment : Temperature
[Haesemeyer et. al., Cell Syst. 2015]
Map of heat responsive neurons
Timescale of heat perception
[Haesemeyer et. al., Neuron 2018]
Zebrafish in the wild : 18-38°C
Exploration of larval zebrafish internal states with temperature
Study the effect of uniform temperature on behavior & brain activity
Define, measure & study internal states
Outline
Introduction
- Behavior
- Setup & larval locomotion
- Thermal modulation of exploratory behaviors
- Calcium imaging
- Spontaneous neural activity : the case of the ARTR
- Data-driven Ising model
Summary & perspectives
Outline
Introduction
-
Behavior
- Setup & larval locomotion
- Thermal modulation of exploratory behaviors
- Calcium imaging
- Spontaneous neural activity : the case of the ARTR
- Data-driven Ising model
Summary & perspectives
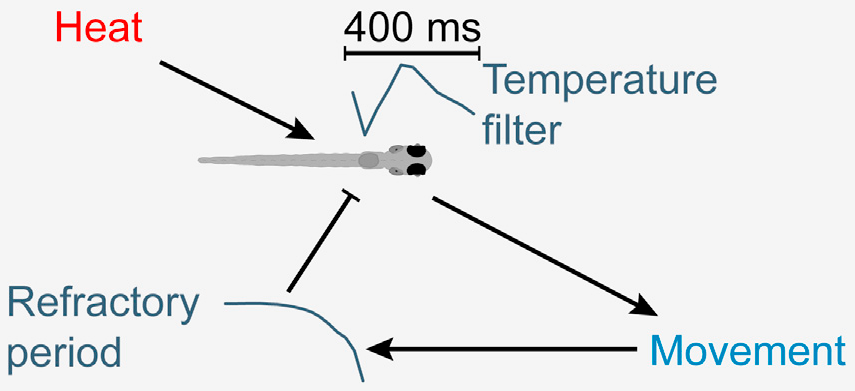
1.1 Setup & larval locomotion

1.1 Setup & larval locomotion
[Gallois & Candelier, PLoS Comp. Biol. 2021]
Offline tracking with FastTrack
Discrete swim bouts
1.1 Larval locomotion
Discrete swim bouts
Short-term kinematic parameters
Interbout interval
1.1 Larval locomotion
Discrete swim bouts
Short-term kinematic parameters
Interbout interval
Displacement
1.1 Larval locomotion
Discrete swim bouts
Short-term kinematic parameters
Interbout interval
Displacement
Turn angle
1.1 Larval locomotion
1.1 Larval locomotion
3 short-term kinematic parameters
Interbout interval
Displacement
Turn amplitude
+ 1 trajectory-based parameter
Turn probability
1.2 Thermal modulation of behavior
[Le Goc et. al., BMC Biol. 2021]

10 batches of 10 fish, 30 minutes
5 temperatures :
T = 18, 22, 26, 30, 33°C
1.2 Parameters are thermally modulated
[Le Goc et. al., BMC Biol. 2021]
1.2 Parameters are thermally modulated
[Le Goc et. al., BMC Biol. 2021]
1.2 Reorientation dynamics
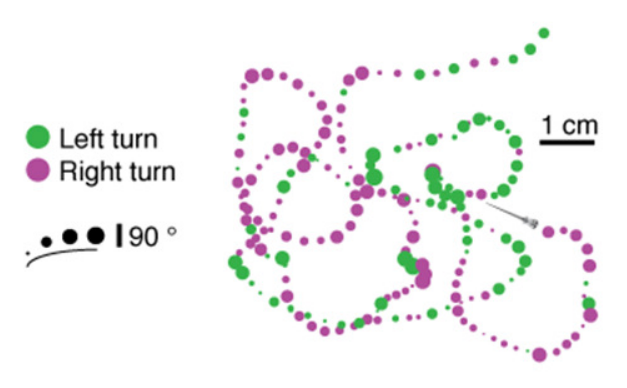
[Dunn, Mu et. al., eLife 2016]
1.2 Orientational persistence
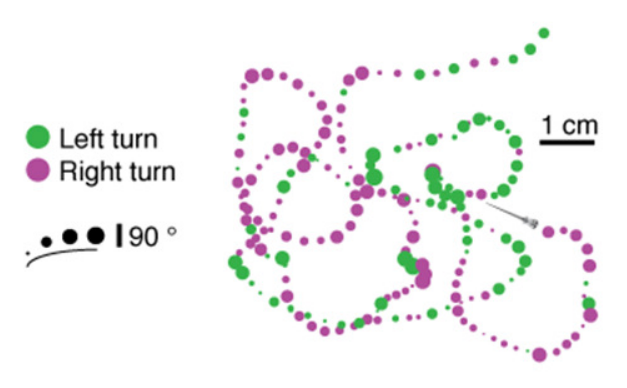
[Dunn, Mu et. al., eLife 2016]
[Karpenko et. al., eLife 2020]
1.2 Orientational persistence
[Le Goc et. al., BMC Biol. 2021]
[Karpenko et. al., eLife 2020]
Ternarized reorientation
Left turn → +1
Forward → 0
Right turn → -1
1.2 Orientational persistence
[Le Goc et. al., BMC Biol. 2021]
[Karpenko et. al., eLife 2020]
Ternarized reorientation
Left turn → +1
Forward → 0
Right turn → -1
1.2 Parameters statistical coupling
[Le Goc et. al., BMC Biol. 2021]
5 thermally modulated parameters :
Sufficient to describe trajectories
1.2 Parameters statistical coupling
[Le Goc et. al., BMC Biol. 2021]
5 thermally modulated parameters :
1.2 Parameters statistical coupling
Nicoguaro CC BY 4.0
1.2 Well-defined behavioral space
[Le Goc et. al., BMC Biol. 2021]
1.2 Behavioral space exploration
[Le Goc et. al., BMC Biol. 2021]
Single-fish experiments, 26°C, 2h long, N = 18
1.2 Inter- & intra-individual variability
[Le Goc et. al., BMC Biol. 2021]
1 color = 1 fish
T = 26°C
1.2 Diffusive modulation of the behavior
[Le Goc et. al., BMC Biol. 2021]
Behavioral modulation time scale ~ 30min
Fish #13 trajectories
1. Summary
Larval zebrafish :
- has a constrained accessible locomotor repertoire
- explores it on a time scale of 30min
- controls the behavioral space
- is a convenient external drive of the internal states
Difficulty to be in a stationnary regime
Temperature :
Outline
Introduction
-
Behavior
- Setup & larval locomotion
- Thermal modulation of exploratory behaviors
-
Calcium imaging
- Spontaneous neural activity : the case of the ARTR
- Data-driven Ising model
Summary & perspectives
2. Calcium imaging
2.1 Spontaneous activity
T = 18, 22, 26, 30, 33°C
30min recordings, ~ 6 volumes/s
~ 50 000 neurons/recordings
2.1 The ARTR
[Dunn, Mu et. al., eLife 2016]
[Wolf et. al., Nat. Commun. 2017]
Anterior Rhombencephalic Turning Region
2.1 The ARTR
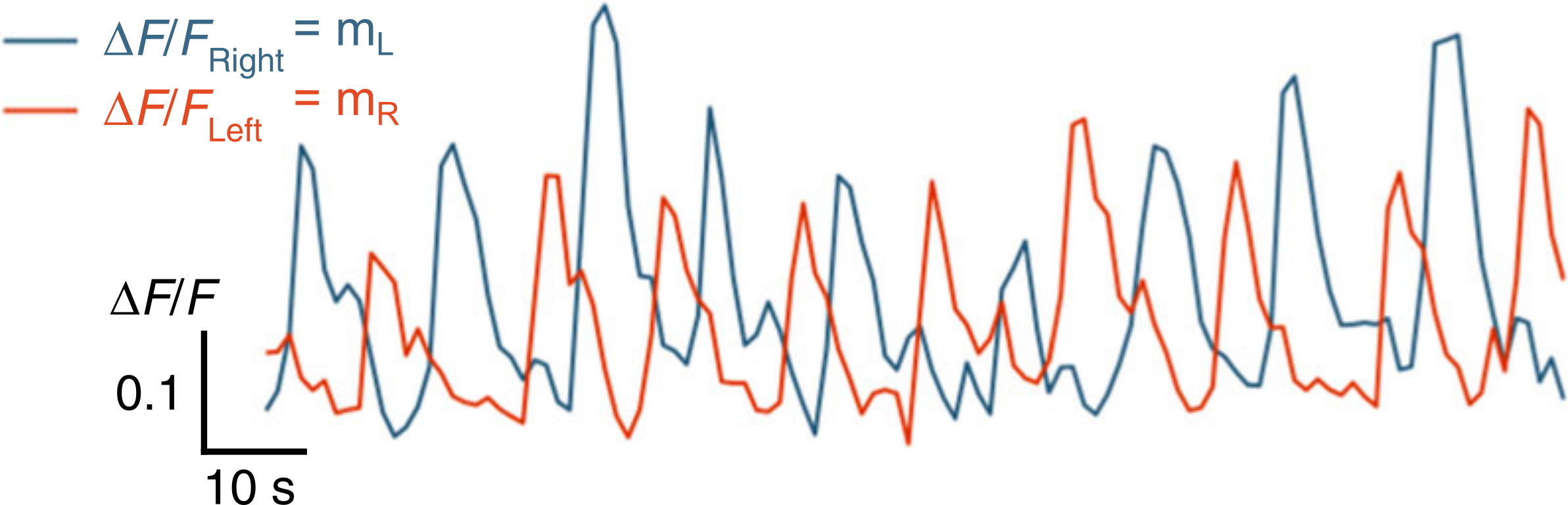
Tuned with turn direction (Dunn, Mu, et. al. eLife 2016)
Tuned with eyes orientation (Wolf, et. al., Nat. Commun. 2017)
[Wolf et. al., Nat. Commun., 2017]
Bilateral circuit with persistent activity
[Wolf et. al., Nat. Commun. 2017]
[Karpenko et. al., eLife 2020]
2.1 Locomotor reorientation dynamics
[Wolf, Le Goc et. al., in prep.]
Random telegraph signal
2.1 ARTR switching dynamics
Behavior
ARTR
[Wolf, Le Goc et. al., in prep.]
2.1 ARTR switching dynamics
Behavior
ARTR
[Wolf, Le Goc et. al., in prep.]
Behavior/Neural activity consistency
2.1 ARTR persistence
[Wolf, Le Goc et. al., in prep.]
With S. Wolf, S. Cocco & R. Monasson (LPENS)
2.2 Data-driven Ising model
[Wolf, Le Goc et. al., in prep.]
?
2.2 Data-driven Ising model
[Schneidman et. al., Nature 2006]
local fields
coupling between neurons i and j
[Wolf, Le Goc et. al., in prep.]
Less constrained model (max. entropy)
2.2 Model parameters inference
[Wolf, Le Goc et. al., in prep.]
2.2 Real vs synthetic data
Real
Synthetic
Monte Carlo Metropolis Hastings sampling of
[Wolf, Le Goc et. al., in prep.]
2.2 Model captures persistence features
[Wolf, Le Goc et. al., in prep.]
2.2 Model captures persistence features
[Wolf, Le Goc et. al., in prep.]
Energy landscape
defines dynamics
2. Summary
ARTR thermal modulation consistent with behavior despite immobilisation
Time-independent, energy-based model reproduces neural dynamics
Persistent activity can emerge from the network
Model captures persistence times variability and thermal modulation
Outline
Introduction
-
Behavior
- Setup & larval locomotion
- Thermal modulation of exploratory behaviors
-
Calcium imaging
- Brain-scale response to water puffs
- Spontaneous neural activity : the case of the ARTR
- Data-driven Ising model
Summary & perspectives
Summary
- Temperature drives locomotor space
- Continuous and diffusive-like modulation of internal states
- Framework to treat inter- & intra-individual variability
- Orientational internal state neural substrate identified : ARTR
- Persistence properties emerge from network
- Interpretable data-driven model captures circuit features
- Find upstream input of ARTR
- Use the model in stimulation context
- Mean-field theory
- Make use of whole-brain data
- Infinite space navigation
- Use internal state as a basis to study reaction to stimulation
Future work
Acknowledgements
Georges Debrégeas
Raphaël Candelier
Volker Bormuth
Ghislaine Morvan-Dubois

Sophia Karpenko
Geoffrey Migault
ulie Lafaye
Natalia Belén Beiza Canelo
Thomas Panier
Benjamin Gallois

Fish facility staff
Abdelkrim Mannioui
Alex Bois
Stéphane Tronche
Marie Breau
Marion Baraban

Rémi Monasson
Simona Cocco
Sébastien Wolf


Hugo Trentesaux
Hippolyte Moulle
Sharbatanu Chatterjee
Mattéo Dommanget-Kott
Alexandre Nauleau
Antoine Hubert
Leonardo Demarchi
Malika Pierrat
Anissa Zerouklane
Acknowledgements
Georges Debrégeas
Raphaël Candelier
Volker Bormuth
Ghislaine Morvan-Dubois



Rémi Monasson
Simona Cocco
Sébastien Wolf
Thanks !



Sophia Karpenko
Geoffrey Migault
ulie Lafaye
Natalia Belén Beiza Canelo
Thomas Panier
Benjamin Gallois
Hugo Trentesaux
Hippolyte Moulle
Sharbatanu Chatterjee
Mattéo Dommanget-Kott
Alexandre Nauleau
Antoine Hubert
Leonardo Demarchi
Malika Pierrat
Anissa Zerouklane
Fish facility staff
Abdelkrim Mannioui
Alex Bois
Stéphane Tronche
Marie Breau
Marion Baraban
1.2 Thermal gradient
10 x 10 fish, 45min
1.2 Temperature preference
1.2 Temperature preference
1.2 Temperature effect on kinematics
1.2 Temperature effect on IBI
1.2 Temperature effect on displacement
1.2 Temperature effect on turn amplitude
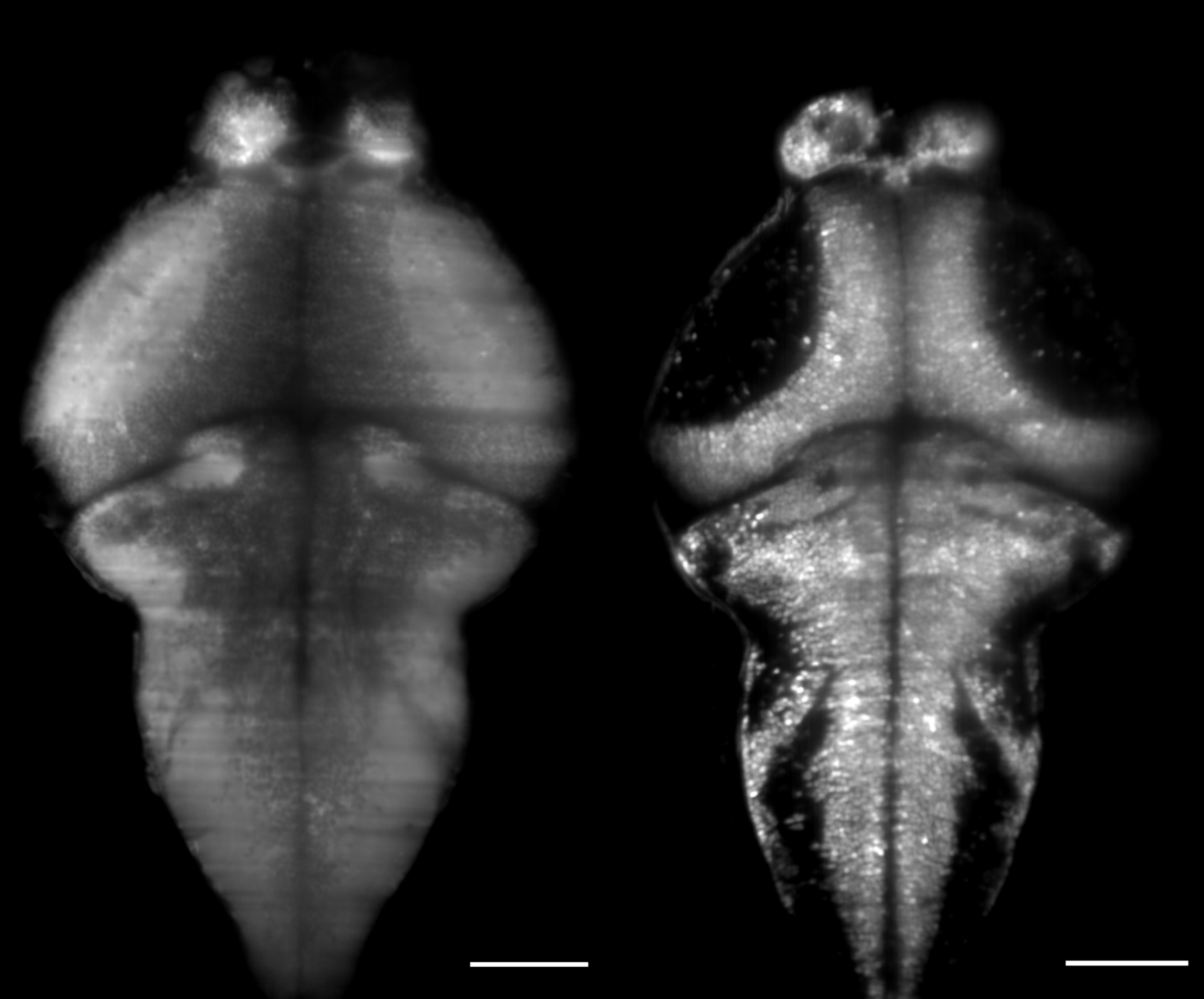
2.1 Response to water puffs
2.1 Response to water puffs
2.1 Response to water puffs
Z-projected trial-averaged mean reponse of 1 fish
hot (n=8)
cold (n=12)
Qualitative response map
2.1 Activity space drives persistence
[Wolf, Le Goc et. al., in prep.]
2.2 Real vs synthetic data
Real
Synthetic
[Wolf, Le Goc et. al., in prep.]
2.2 Real vs synthetic data
[Wolf, Le Goc et. al., in prep.]